Uranium Enrichment
On this page:
Uranium Isotopes
When uranium is mined, it consists of approximately 99.3% uranium-238 (U238), 0.7% uranium-235 (U235), and < 0.01% uranium-234 (U234). These are the different uranium isotopes. Isotopes of uranium contain 92 protons in the atom's center or nucleus. (The number of protons in the nucleus is what makes the atoms "uranium.") The U238 atoms contain 146 neutrons, the U235 atoms contain 143 neutrons, and the U234 atoms contain only 142 neutrons. The total number of protons plus neutrons gives the atomic mass of each isotope — that is 238, 235, or 234, respectively. On an atomic level, the size and weight of these isotopes are slightly different. This implies that with the right equipment and under the right conditions, the isotopes can be separated.
Enriching Uranium
The nuclear fuel used in a nuclear reactor needs to have a higher concentration of the U235 isotope than that which exists in natural uranium ore. U235 when concentrated (or "enriched") is fissionable in light-water reactors (the most common reactor design in the USA). During fission, the nucleus of the atom splits apart producing both heat and extra neutrons. Under controlled conditions, these extra neutrons can cause additional, nearby atoms to fission and a nuclear reaction can be sustained. The heat energy released, by the controlled nuclear reaction within the nuclear reactor, can be harnessed to produce electricity. Commercially, the U235 isotope is enriched to 3 to 5% (from the natural state of 0.7%) and is then further processed to create nuclear fuel.
At the conversion plant, uranium oxide is converted to the chemical form of uranium hexafluoride (UF6) to be usable in an enrichment facility. UF6 is used for a couple reasons; 1) The element fluorine has only one naturally-occurring isotope which is a benefit during the enrichment process (e.g. while separating U235 from U238 the fluorine does not contribute to the weight difference), and 2) UF6 exists as a gas at a suitable operating temperature.
The two primary hazards at enrichment facilities include chemical hazards that could be created from a UF6 release and criticality hazards associated with enriched uranium.
There are several enrichment processes utilized worldwide. They are:
Gaseous Diffusion
|
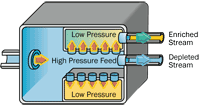
The gaseous diffusion process uses molecular diffusion to separate a gas from a two-gas mixture. The isotopic separation is accomplished by diffusing uranium [which has been combined with fluorine to form uranium hexafluoride (UF6) gas] through a porous membrane (barrier), and using the different molecular velocities of the two isotopes to achieve separation. |
Gaseous diffusion was the first commercial process used in the United States to enrich uranium. These facilities utilized massive amounts of electricity and as the centrifuge technology matured the existing gaseous diffusion plants became obsolete. Worldwide they have all been replaced by second-generation technology, which requires far less electric power to produce equivalent amounts of separated uranium. These facilities are now considered obsolete.
Process: In a gaseous diffusion enrichment plant, uranium hexafluoride (UF6) gas was fed into the plant's pipelines where it was pumped through special filters called barriers or porous membranes. The holes in the barriers were so small that there was barely enough room for the UF6 gas molecules to pass through as shown in this diagram. The isotope enrichment occurred because the lighter UF6 gas molecules (with the U234 and U235 atoms) diffused faster through the barriers than the heavier UF6 gas molecules containing U238. One barrier wasn't enough though. It took many hundreds of barriers, one after the other, before the UF6 gas contained enough U235 to be used in nuclear fuel. At the end of the process, the enriched UF6 gas was withdrawn from the pipelines and condensed back into a liquid that was then poured into containers. The UF6 was allowed to cool and solidify before it was transported to fuel fabrication facilities. The diagram to the right illustrates the gaseous diffusion enrichment process.
Legacy Plants: The gaseous diffusion plants that were historically operated in the United States were located in Paducah, Kentucky; Piketon, Ohio; and Oak Ridge, Tennessee. The U.S. government leased the Piketon and Paducah plants to the United States Enrichment Corporation (USEC) in March 4, 1997. Both plants enriched uranium for commercial use and were regulated by the Nuclear Regulatory Commission (NRC). Since then, both plants have been shut down. The certificates were transferred back to the U.S. Department of Energy (DOE); the agency responsible for the safe decommissioning of the plants.
While the two gaseous diffusion plants were under NRC purview, their certificates were issued in accordance with 10 CFR Part 76. The NRC maintained one or two resident inspectors on site while the facilities were in operation. Inspectors out of the regional and headquarters office also performed inspections. Documents pertaining to safety inspections conducted by the NRC can be found below. The current status of decommissioning can be obtained on the Portsmouth/Paducah Project Office page maintained by the DOE.
Paducah
Portsmouth
Both the operation and decommissioning of the gaseous diffusion plant in Oak Ridge, TN was regulated by the DOE and was never under NRC purview.
|
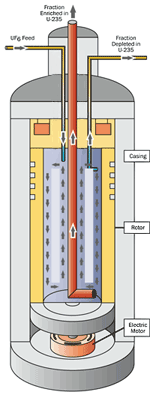
The gas centrifuge process uses a large number of rotating cylinders in series and parallel configurations. Gas is introduced and rotated at high speed, concentrating the component of higher molecular weight toward the outer wall of the cylinder and the lower molecular weight component toward the center. The enriched and the depleted gases are removed by scoops. |
Gas Centrifuge
Gas centrifuge enrichment is the current process by which commercial enrichment is being performed in the United States. UF6 gas is placed in a gas centrifuge cylinder and rotated at a high speed. This rotation creates a strong centrifugal force so that the heavier gas molecules (UF6 containing U238 atoms) move towards the outside of the cylinder. The lighter gas molecules (containing U235) collect closer to the center. The stream that is slightly enriched in U235 is withdrawn and fed into the next centrifuge; the next higher stage. The slightly depleted stream (with a lower concentration of U235) is recycled back into the next lower stage.
A gas centrifuge facility contains long lines of many rotating cylinders. These cylinders are connected in both series and parallel formations. Centrifuge machines are interconnected to form trains and cascades. At the final withdrawal point, the UF6 is enriched to the desired amount.
Currently, the only gas centrifuge commercial production plant is the URENCO USA (UUSA) facility licensed as Louisiana Energy Services (LES). UUSA is currently operating in Eunice, NM. Two other licenses were granted by the NRC for the construction of commercial gas centrifuge facilities. The status of these licensees are found in this chart.
Laser Separation
The laser separation technology is under development for possible use to enrich uranium. Uranium can be enriched by separating isotopes of uranium with lasers. Molecules can be excited by laser light; this is called photoexcitation. Lasers can increase the energy in the electrons of a specific isotope, changing its properties and allowing it to be separated.
In general, the enrichment process entails using three major systems, which are the laser systems, optical systems, and separation module system. Tunable lasers can be developed to deliver a highly monochromatic light (light of a single-color). The light from these lasers can photo-ionize a specific isotopic species while not affecting other isotopic species. The affected species is then chemically changed, which enables the material to be separated. The laser separation technology developed by DOE used a uranium metal alloy as its feed material, while the Separation of Isotopes by Laser Excitation (SILEX) method uses UF6 as the feed material.
No laser separation uranium enrichment plants are currently operating in the United States. However, on May 12, 2008, the NRC approved a license amendment submitted by General Electric - Hitachi seeking approval for research and development associated with laser enrichment to be conducted at its Global Nuclear Fuels-Americas, LLC facility in Wilmington, NC. GE Hitachi constructed a test loop and illustrated the SILEX laser isotope separation process. On September 25, 2012, NRC staff issued a construction and operating license for the facility. The current status of the facility can be found on the New Fuel Cycle Facility Licensing page.
Page Last Reviewed/Updated Tuesday, May 26, 2026
Page Last Reviewed/Updated Tuesday, May 26, 2026