Classroom Activity

Unit 1: Radiation
How Can You See the Footprints of Radiation?

While radiation cannot be seen, the cloud chamber allows you to see the tracks it leaves in a dense gas.
Materials
- small transparent container with transparent lid
- flat black spray paint
- blotter paper
- pure ethyl alcohol
- radioactive source
- masking tape
- dry ice
- styrofoam square
- flashlight
- gloves or tongs to handle the dry ice
First, paint the bottom of the container with black paint and let it dry. Then cut the blotter paper into a strip about as wide as the height of the container. Cut two windows in the strip, as shown, and place it against the inside of the container.
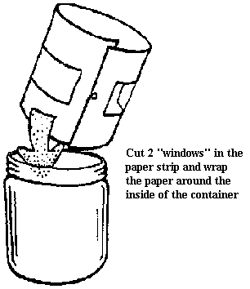
Pour enough ethyl alcohol into the cloud chamber to cover the bottom of the container. The blotter paper will absorb most of it.
Place the radioactive source in the cloud chamber and seal the lid with tape.
Place the cloud chamber on the dry ice to super-chill it. Wait about five minutes. Darken the room. Shine the flashlight through the windows of the chamber while looking through the lid. You should see "puffs" and "trails" coming from the source. These are the "footprints" of radiation as it travels through the alcohol vapor. The vapor condenses as the radiation passes through. This is much like the vapor trail left by high flying jets.
Do you see radiation in the cloud chamber?
Other Ideas To Explore
Try to identify these footprints:
- Alpha: sharp tracks about 1 cm long
- Beta: thin tracks 3 cm to 10 cm long
- Gamma: faint, twisting and spiraling tracks
Caution: Dry ice should be handled very carefully! It can burn unprotected skin.
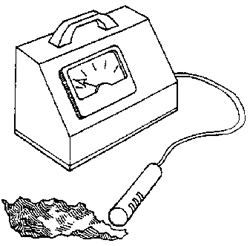
How radioactive are different materials?
Materials
- Geiger counter
- Radioactive sources such as:
- cloisonne jewelry
- commercially available source from a science supply house
- luminescent clock face
- Shielding materials such as:
- paper
- aluminum foil
- brick
- jar of water
- piece of wood
- glass pane
- sheet of lead
Directions:
- One at a time, test each item that is a source of radioactivity by placing the source 2 inches from the Geiger counter probe. Which item has the highest reading?
The lowest? - Place the radioactive source that had the highest reading 2 inches from the Geiger counter probe. One at a time, test each of your shielding materials by placing them between the source and the counter. Do you think the density of the shield is important?
Why?
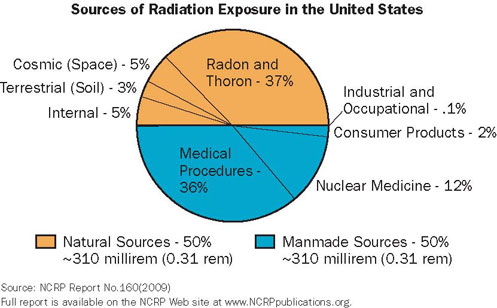 |
| This chart shows that natural sources of radiation account for about 50% of all public exposure while man-made sources account for the remaining 50%. |
We live in a radioactive world and always have. Radiation is all around us as a part of our natural environment. It is measured in millirems (mrems). The annual average dose per person from all sources is about 620 mrems, but it is not uncommon for any of us to receive more than that in a given year (largely due to medical procedures).
To find your average annual dose (mrems), use the interactive Personal Annual Radiation Dose Calculator or this printer friendly worksheet.
Unit 3: Nuclear Reactors/Energy Generation
A Day in the Life of an NRC Resident Inspector
Page Last Reviewed/Updated Wednesday, November 15, 2023
Page Last Reviewed/Updated Wednesday, November 15, 2023

