Resolution of Generic Safety Issues: Introduction ( NUREG-0933, Main Report with Supplements 1–35 )
The U.S. Nuclear Regulatory Commission (NRC), through its assessment of plant operations, identifies potentially significant issues involving public health and safety, the common defense and security, or the environment that could affect multiple entities under NRC jurisdiction. The NRC uses the Generic Issues (GI) Program to document and track the resolution of such issues. The GI Program also reports the GIs' status and resolutions to Congress and the public. The resolution of GIs may involve developing new or revised rules or guidance, or interpreting rules or guidance, that affect nuclear power plant licensees, nuclear material certificate holders, or holders of other regulatory approvals. Congress requires that the NRC maintain such a program per Section 210 of the Energy Reorganization Act of 1974 (Public Law 95-209).
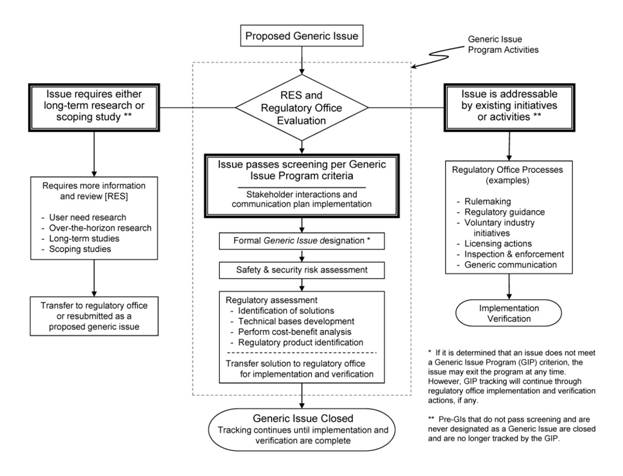
The NRC staff or members of the public may propose a GI when issues are identified that indicate there might be weaknesses in NRC regulations that ensure public health and safety and security for nuclear matters. However, the GI Program will address only those issues that meet the following seven criteria:
- The issue affects public health and safety, the common defense and security, or the environment (with respect to radiological health and safety).
- The issue applies to two or more facilities, licensees, certificate holders, or holders of other regulatory approvals.
- The issue is not being addressed through other regulatory programs and processes or existing regulations, policies, or guidance.
- The issue can be resolved by a new or revised regulation, policy, or guidance.
- The issue's risk or safety significance can be adequately determined in a timely manner.
- The issue is well defined, discrete, and technical.
- Resolution of the issue may involve review, analysis, or action by the affected licensees, certificate holders, or holders of other regulatory approvals.
The NRC describes its process for resolving GIs in Management Directive 6.4, “Generic Issues Program,” 1868 and Office of Nuclear Regulatory Research Office Instruction TEC-002, “Procedure for Processing Generic Issues.” 2216 Appendix G to this report documents the current GI Program and the history of changes. These documents reflect program improvement initiatives. The current process includes three distinct stages: Screening, Assessment, and Regulatory Office Implementation. During each stage, the NRC staff evaluates whether the issue is significant enough to proceed to the next stage or recommends that the issue exit the GI Program. When issues exit the GI Program, the possible outcomes include no action, further research, or the transfer of responsibility to an appropriate regulatory program or possibly an industry initiative. The GI Program provides feedback about the outcome of each stage to the person proposing the GI (requestor), the public, and the appropriate regulatory office. Issues that proceed through all three stages result in regulatory solutions that are derived by the regulatory offices for implementation and verification. The figure below presents an overview of the GI Program.
On the Generic Issue Dashboard page of the NRC's public Web site, https://www.nrc.gov/about-nrc/regulatory/gen-issues/dashboard.html, the staff and members of the public can view progress towards resolving GIs that the NRC has identified for regulation and guidance development. The NRC also publishes a semiannual Generic Issue Management Control System (GIMCS) report, which is available in the NRC's Public Document Room or from the Publicly Available Records component of the Agencywide Documents Access and Management System (ADAMS). Additionally, the staff uses NUREG-0933 to document the resolutions of all resolved GIs and the assessments of work remaining on unresolved GIs. The GIMCS report indicates the GIs identified since the last publication of NUREG-0933.


