Backgrounder on Medical Use of Radioactive Materials
Radioactive materials can be helpful to diagnose and treat illnesses or in medical research. The Nuclear Regulatory Commission regulates these uses. NRC rules aim to ensure radioactive materials are used properly and in a way that protects patients, medical workers, the public and the environment.
About one-third of all patients admitted to hospitals are diagnosed or treated using radiation or radioactive materials. This branch of medicine is called nuclear medicine. The radioactive materials used are called radiopharmaceuticals.
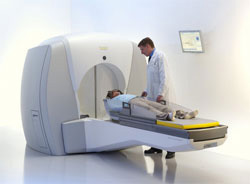
Gamma Knife® (Courtesy: Elekta)
|
For diagnosis, a small amount of material is injected, inhaled or swallowed. The material collects in the area being studied, where it emits photons. These photons can be seen by a device known as a gamma camera. Images from the camera can show the organ's function and composition. They can help physicians find, identify and measure tumors, or view problems in an organ. For example, technetium-99m is used to diagnose bone, heart or other organ problems. Radioactive iodine is used in imaging the thyroid gland.
For therapy, radioactive materials are used to kill cancerous tissue, shrink a tumor or reduce pain. There are three main types of therapy in nuclear medicine.

Brachytherapy Seeds
(Courtesy: Oak Ridge Associated Universities) |
Teletherapy targets cancerous tissue with an intense beam of radiation. One example uses a device called the Gamma Knife®, which focuses radiation from a number of cobalt-60 sources on a spot deep within brain tissue.
Brachytherapy uses lower-activity sources sealed in "seeds" that are placed close to or within cancerous tissue, such as in the breast, prostate or cervix. The seeds are injected or surgically implanted, then removed after the patient receives the prescribed dose. Intravascular brachytherapy systems place small sources into arteries using catheters.
Therapeutic nuclear medicine uses high doses of radiation from materials that are injected into or ingested by the patient. For example, radioactive iodine can destroy or shrink a diseased thyroid.
Who Regulates
Federal, state and local agencies share responsibility for overseeing the uses of radiation in medicine. Radioactive material is regulated by the NRC or an Agreement State. Agreement States have signed agreements with the NRC allowing them to regulate the use of certain radioactive materials. These states issue licenses to and oversee medical users such as university medical centers, hospitals, clinics and doctors in private practice. The NRC similarly licenses and oversees medical uses in the non-Agreement States, the District of Columbia, Puerto Rico and other U.S. territories. The NRC is also responsible for ensuring the common defense and security of nuclear materials.
The Food and Drug Administration reviews the safety and use of radiopharmaceuticals and machines, such as x-rays, that produce radiation but do not make or use radioactive material. But it is the states, not the FDA, that actually regulate their use.
To clarify each agency's role, the NRC and the FDA have a Memorandum of Understanding. Both agencies have liaison officers and key managers and technical personnel who coordinate responses to emergencies or specific events of mutual interest. These officials have carried out joint inspections to better understand medical events involving device failures and human- or computer-generated errors. Senior managers from both agencies also meet each year.
How the NRC Regulates
The NRC and before it, the Atomic Energy Commission, have regulated the medical use of radioactive materials since 1946. The regulations are laid out in Title 10 of the Code of Federal Regulations, Part 35. In developing and implementing these regulations, the NRC follows its Medical Use Policy Statement. The policy says the NRC will:
- regulate the uses of radionuclides in medicine to provide for the safety of workers and the public;
- not intrude into medical judgments affecting patients, except as necessary to provide for the safety of workers and the public;
- regulate radiation safety primarily to ensure that the doctor’s directions are carried out, when justified by the risk to the patient; and
- consider industry and professional standards for acceptable approaches to radiation safety.
The NRC and Agreement States license facilities, authorize individuals to administer radioactive materials, and develop regulations and guidance. The regulations also provide for inspections, investigations and enforcement programs to ensure the safety of medical uses.
The NRC relies on a committee of medical experts for advice. The Advisory Committee on the Medical Uses of Isotopes meets twice a year to discuss with NRC staff current practices and trends in medical uses of radioactive materials. ACMUI members include a nuclear cardiologist, a nuclear medicine physician, two radiation oncologists, an Agreement State representative, a nuclear pharmacist, a therapy medical physicist, a medical physicist in nuclear medicine, a patients' rights advocate, a health care administrator, an FDA representative and a radiation safety officer.
From time to time, the NRC revises it regulations. There have been some major revisions that affect medical uses in recent years.
- In 2002, the NRC adopted revisions to focus on the procedures that pose the highest risk and the requirements that are essential for protecting patient safety.
- In 2005, the NRC amended the training and validation requirements for doctors authorized to use radioactive materials and revised the requirements for recognizing specialty boards.
- In 2019, the NRC amended its medical use regulations for medical events and training and experience requirements.
More information on medical uses of radioactive materials can be found on the NRC's website.
September 2020
Page Last Reviewed/Updated Tuesday, September 22, 2020
Page Last Reviewed/Updated Tuesday, September 22, 2020

