Schedules
§ 30.70 Schedule A--Exempt concentrations.
[See footnotes at the end of this table]
| Element (atomic number) | Isotope | Col. I | Col. II |
|---|---|---|---|
| Gas Concentration μCi/ml1 | Liquid and Solid Concentration μCi/ml2 | ||
| Antimony (51) | Sb 122 | 3 x 10-4 | |
| Sb 124 | 2 x 10-4 | ||
| Sb 125 | 1 x 10-3 | ||
| Argon (18) | A 37 | 1 x 10 -3 | |
| A 41 | 4 x 10 -7 | ||
| Arsenic (33) | As 73 | 5 x 10-3 | |
| As 74 | 5 x 10-4 | ||
| As 76 | 2 x 10-4 | ||
| As 77 | 8 x 10-4 | ||
| Barium (56) | Ba 131 | 2 x 10-3 | |
| Ba 140 | 3 x 10-4 | ||
| Beryllium (4) | Be 7 | 2 x 10-2 | |
| Bismuth (83) | Bi 206 | 4 x 10-4 | |
| Bromine (35) | Br 82 | 4 x 10-7 | 3 x 10-3 |
| Cadmium (48) | Cd 109 | 2 x 10-3 | |
| Cd 115M | 3 x 10-4 | ||
| Cd 115 | 3 x 10-4 | ||
| Calcium (20) | Ca 45 | 9 x 10-5 | |
| Ca 47 | 5 x 10-4 | ||
| Carbon (6) | C 14 | 1 x 10 -6 | 8 x 10-3 |
| Cerium (58) | Ce 141 | 9 x 10-4 | |
| Ce 143 | 4 x 10-4 | ||
| Ce 144 | 1 x 10-4 | ||
| Cesium (55) | Cs 131 | 2 x 10-2 | |
| Cs 134m | 6 x 10-2 | ||
| Cs 134 | 9 x 10-5 | ||
| Chlorine (17) | Cl 38 | 9 x 10-7 | 4 x 10 -3 |
| Chromium (24) | Cr 51 | 2 x 10-2 | |
| Cobalt (27) | Co 57 | 5 x 10 -3 | |
| Co 58 | 1 x 10 -3 | ||
| Co 60 | 5 x 10-4 | ||
| Copper (29) | Cu 64 | 3 x 10-3 | |
| Dysprosium (66) | Dy 165 | 4 x 10-3 | |
| Dy 166 | 4 x 10-4 | ||
| Erbium (68) | Er 169 | 9 x 10-4 | |
| Er 171 | 1 x 10-3 | ||
| Europium (63) | Eu 152 (T/2=9.2 hrs) | 6 x 10-4 | |
| Eu 155 | 2 x 10-3 | ||
| Fluorine (9) | F 18 | 2 x 10-6 | 8 x 10-3 |
| Gadolinium (64) | Gd 153 | 2 x 10-3 | |
| Gd 159 | 8 x `0-4 | ||
| Gallium (31) | Ga 72 | 4 x 10-4 | |
| Germanium (32) | Ge 71 | 2 x 10-2 | |
| Gold (79) | Au 196 | 2 x 10 -3 | |
| Au 198 | 5 x 10-4 | ||
| Au 199 | 2 x 10 -3 | ||
| Hafnium (72) | Hf 181 | 7 x 10-4 | |
| Hydrogen (1) | H 3 | 5 x 10-6 | 3 x 10-2 |
| Indium (49) | In 113M | 1 x 10 -2 | |
| In 114M | 2 x 10-4 | ||
| Iodine (53) | I 126 | 3 x 10-9 | 2 x 10-5 |
| I 131 | 3 x 10-9 | 2 x 10-5 | |
| I 132 | 8 x 10-8 | 6 x 10-4 | |
| I 133 | 1 x 10-8 | 7 x 10-5 | |
| I 134 | 2 x 10-7 | 1 x 10-3 | |
| Iridium (77) | Ir 190 | 2 x 10-3 | |
| Ir 192 | 4 x 10-4 | ||
| Ir 194 | 3 x 10-4 | ||
| Iron (26) | Fe 55 | 8 x 10-3 | |
| Fe 59 | 6 x 10-4 | ||
| Krypton (36) | Kr 85M | 1 x 10-6 | |
| Kr 85 | 3 x 10 -6 | ||
| Lanthanum (57) | La 140 | 2 x 10-4 | |
| Lead (82) | Pb 203 | 4 x 10-3 | |
| Lutetium (71) | Lu 177 | 1 x 10-3 | |
| Manganese (25) | Mn 52 | 3 x 10-4 | |
| Mn 54 | 1 x 10-3 | ||
| Mn 56 | 1 x 10-3 | ||
| Mercury (80) | Hg 197M | 2 x 10-3 | |
| Hg 197 | 3 x 10-3 | ||
| Hg 203 | 2 x 10-4 | ||
| Molybdenum (42) | Mo 99 | 2 x 10-3 | |
| Neodymium (60) | Nd 147 | 6 x 10-4 | |
| Nd 149 | 3 x 10-3 | ||
| Nickel (28) | Ni 65 | 1 x 10-3 | |
| Niobium (Columbium) (41) | Nb 95 | 1 x 10-3 | |
| Nb 97 | 9 x 10-3 | ||
| Osmium (76) | Os 185 | 7 x 10-4 | |
| Os 191M | 3 x 10-2 | ||
| Os 191 | 2 x 10-3 | ||
| Os 193 | 6 x 10-4 | ||
| Palladium (46) | Pd 103 | 3 x 10-3 | |
| Pd 109 | 9 x 10-4 | ||
| Phosphorus (15) | P 32 | 2 x 10-4 | |
| Platinum (78) | Pt 191 | 1 x 10-3 | |
| Pt 193M | 1 x 10-2 | ||
| Pt 197M | 1 x 10-2 | ||
| Pt 197 | 1 x 10-3 | ||
| Potassium (19) | K 42 | 3 x 10-3 | |
| Praseodymium (59) | Pr 142 | 3 x 10-4 | |
| Pr 143 | 5 x 10-4 | ||
| Promethium (61) | Pm 147 | 2 x 10-3 | |
| Pm 149 | 4 x 10-4 | ||
| Rhenium (75) | Re 183 | 6 x 10-3 | |
| Re 186 | 9 x 10-4 | ||
| Re 188 | 6 x 10-4 | ||
| Rhodium (45) | Rh 103M | 1 x 10-1 | |
| Rh 105 | 1 x 10-3 | ||
| Rubidium (37) | Rb 86 | 7 x 10-4 | |
| Ruthenium (44) | Ru 97 | 4 x 10-4 | |
| Ru 103 | 8 x 10-4 | ||
| Ru 105 | 1 x 10-3 | ||
| Ru 106 | 1 x 10-4 | ||
| Samarium (62) | Sm 153 | 8 x 10-4 | |
| Scandium (21) | Sc 46 | 4 x 10-4 | |
| Sc 47 | 9 x 10-4 | ||
| Sc 48 | 3 x 10-4 | ||
| Selenium (34) | Se 75 | 3 x 10-3 | |
| Silicon (14) | Si 31 | 9 x 10-3 | |
| Silver (47) | Ag 105 | 1 x 10-3 | |
| Ag 110M | 3 x 10-4 | ||
| Ag 111 | 4 x 10-4 | ||
| Sodium (11) | Na 24 | 2 x 10-3 | |
| Strontium (38) | Sr 85 | 1 x 10-4 | |
| Sr 89 | 1 x 10-4 | ||
| Sr 91 | 7 x 10-4 | ||
| Sr 92 | 7 x 10-4 | ||
| Sulfur (16) | S 35 | 9 x 10 -8 | 6 x 10-4 |
| Tantalum (73) | Ta 182 | 4 x 10-4 | |
| Technetium (43) | Tc 96M Tc 96 | 1 x 10-1 1 x 10-3 | |
| Tellurium (52) | Te 125M | 2 x 10-3 | |
| Te 127M | 6 x 10-4 | ||
| Te 127 | 3 x 10-3 | ||
| Te 129M | 3 x 10-4 | ||
| Te 131M | 6 x 10-4 | ||
| Te 132 | 3 x 10-4 | ||
| Terbium (65) | Tb 160 | 4 x 10-4 | |
| Thallium (81) | Tl 200 Tl 201 Tl 202 Tl 204 | 4 x 10-3 3 x 10-3 1 x 10-3 1 x 10-3 | |
| Thulium (69) | Tm 170 Tm 171 | 5 x 10-4 5 x 10-3 | |
| Tin (50) | Sn 113 Sn 125 | 9 x 10-4 2 x 10-4 | |
| Tungsten (Wolfram) (74) | W 181 W 187 | 4 x 10-3 7 x 10-4 | |
| Vanadium (23) | V 48 | 3 x 10-4 | |
| Xenon (54) | Xe 131M | 4 x 10-6 | |
| Xe 133 | 3 x 10-6 | ||
| Xe 135 | 1 x 10-6 | ||
| Ytterbium (70) | Yb 175 | 1 x 10-3 | |
| Yttrium (39) | Y 90 | 2 x 10-4 | |
| Y 91M | 3 x 10-2 | ||
| Y 91 | 3 x 10-4 | ||
| Y 92 | 6 x 10-4 | ||
| Y 93 | 3 x 10-4 | ||
| Zinc (30) | Zn 65 | 1 x 10-3 | |
| Zn 69M | 7 x 10-4 | ||
| Zn 69 | 2 x 10-2 | ||
| Zirconium (40) | Zr 95 | 6 x 10-4 | |
| Zr 97 | 2 x 10-4 | ||
| Beta and/or gamma emitting byproduct not listed above with half-life less than three years | 1 x 10-10 | 1 x 10-6 |
NOTE 1: Many radioisotopes disintegrate into isotopes which are also radioactive. In expressing the concentrations in Schedule A, the activity stated is that of the parent isotope and takes into account the daughters.
NOTE 2: For purposes of 30.14 where there is involved a combination of isotopes, the limit for the combination should be derived as follows: Determine for each isotope in the product the ratio between the concentration present in the product and the exempt concentration established in Schedule A for the specific isotope when not in combination. The sum of such ratios may not exceed "1" (i.e., unity).
Example:
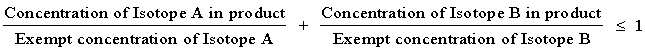
[30 FR 8185, June 26, 1965, as amended at 35 FR 3982, Mar. 3, 1970; 38 FR 29314, Oct. 24, 1973; 59 FR 5520, Feb. 7, 1994]
Page Last Reviewed/Updated Tuesday, August 29, 2017
Page Last Reviewed/Updated Tuesday, August 29, 2017

