Information Notice No. 99-02: Guidance to Users on the Implementation of a New Single-Source Dose-Calculation Formalism and Revised Air-Kerma Strength Standard for Iodine-125 Sealed Sources
UNITED STATES
NUCLEAR REGULATORY COMMISSION
OFFICE OF NUCLEAR MATERIAL SAFETY AND SAFEGUARDS
WASHINGTON, D.C. 20555
January 21, 1999
| NRC INFORMATION NOTICE 99-02: |
GUIDANCE TO USERS ON THE IMPLEMENTATION OF A NEW SINGLE-SOURCE DOSE-CALCULATION FORMALISM AND REVISED AIR-KERMA STRENGTH STANDARD FOR IODINE-125 SEALED SOURCES |
Addressees:
All medical licensees authorized to conduct brachytherapy treatments
Purpose:
The U.S. Nuclear Regulatory Commission (NRC) is issuing this information notice to alert addressees to two changes affecting Iodine-125 sealed source dosimetry that, if not properly implemented, may result in unintended differences in doses delivered to patients. These changes are: (1) the American Association of Physicists in Medicine (AAPM) Task Group 43 (TG43) dosimetry protocol; IN 99-02
Description of Circumstances:
It is important for all licensees performing radiation therapy with interstitially implanted I-125 seeds to be aware of two recent changes impacting the calculations of dose delivered from such implants. Chronologically, the first of these changes
occurred in 1995, when the AAPM issued its TG43 report on interstitial brachytherapy dosimetry. This report establishes a new dose-calculation formalism that yields calculated dose rates, for the same I-125 seed, that are smaller by 10-18% than those obtained previously.
The second change, which is to be implemented on January 1, 1999, is a revision of the National Institute of Standards and Technology (NIST) air-kerma strength (SK) standard for low-energy interstitial brachytherapy seeds, This revision reduces the stated air-kerma strength by 11.5 percent from that of the presently used value established by NIST in 1985.
Discussion
While optional, conversion to the new TG43 dosimetry protocol (described in Attachment 1) is strongly recommended. The long term consequences of not converting to the TG43 protocol could lead to significant errors in patient doses. For example, physicians in practices that have not adopted the new protocol, must consistently convert all recommended dose prescriptions derived from literature and other sources, that now use the TG43 protocol, to their own "unique" dosimetry protocol. The chances for the introduction of dose prescription errors from this conversion is expected to be very high. This source of potential prescription errors can be eliminated by simply converting to the recommended TG43 dosimetry protocol, as described in Attachment 1.
Physicists should not implement revised dose prescription guidelines nor implement TG43 dose calculations without consulting with the responsible radiation oncologist. Guidance on implementing numerically equivalent TG43 dose calculations on treatment planning systems is also given in Attachment 1.
The scheduled change in I-125 seed calibration values, for implementation by NIST, Accredited Dosimetry Calibration Laboratories, and source (seed) suppliers on or about January 1, 1999, requires that the dose-rate constant presently being used be updated to a new value that incorporates the 1999 NIST change in the air-kerma value. A status report from NIST on the change over to the new I-125 seed calibration values was received, on January 5, 1999, via Email from Dr. Bert Coursey (Attachment 2), Although source vendors are expected to also convert to the 1999 air-kerma strength standard on January 1, 1999, there is no guarantee this will happen as scheduled (see question 1 in Attachment 2). Thus, during this transition period from the old (1985) air-kerma standard to the new (1999) standard, licensees need to be very vigilant as to which standard for air-kerma strength was used by the vendor suppling their I-125 seed sources.
If licensees do not make the necessary adjustments, as described in Attachment 1 for both those practices that have adopted the TG43 dosimetry protocol and those that have not, the doses delivered to patients will be increased by 11.5 percent, without the knowledge of the authorized user physician. Also, the NIST revision of the air-kerma standard will require a corresponding adjustment to the calibration factor(s) used with source calibration instruments, as described in Attachment 1.
The essential point is that correct implementation of the TG43 formalism or the NIST revised air-kerma strength standard, either singly or in combination, does not change the quantity of radioactive material from that used previously to achieve the desired clinical outcome. The new TG43 formalism and revised air-kerma standard, simply provide a more accurate and rigorous estimate of the actual dose delivered to the prescription point(s). If an institution erroneously adjusts the prescribed doses upward to achieve the same numerical prescribed dose used before adopting the new TG43 formalism and/or the revised air-kerma standard the administered dose could differ by upwards of 30 percent from the dose administered in pre TG43 and/or revised Air-Kerma practice. Such errors could lead to undesired clinical outcomes and possibly patient misadministrations as defined in 10 CFR Part 35.
If you have questions on implementing the TG43 dosimetry recommendations or adapting to the revised source strength standard, please contact the Radiological Physics Center (RPC) at MD Anderson Cancer Center, Houston, TX at (713)-792-3233.
If you encounter adverse events or product problems in using I-125 sources, produced after January 1, 1999, you are encouraged to report these to MedWatch, the Food and Drug Administration's (FDA) voluntary reporting program. You may report to MedWatch by phone at 1-800-FDA-1088; by FAX at 1-800-FDA-0178; over the Internet at http://www.fda.gov/medwatch/; or by mail to MedWatch, Food and Drug Administration, 5600 Fishers Lane (HF-2), Rockville, MD 20857. More serious events that result in death, serious illness or injuries require reporting to the FDA under the Safe Medical Devices Act of 1990.
This information notice requires no specific action nor written response. If you have any questions about the information in this notice, please contact the technical contact listed below or the appropriate NRC regional office.
The NRC staff would like to express their appreciation to the members of the AAPM Ad Hoc Subcommittee on Low-Energy Seed Dosimetry, particularly the Chair, Jeffrey F. Williamson, Ph.D., for their time and effort in writing Attachment 1 to this Information Notice.
| |
/s/'d by
Donald A. Cool, Director
Division of Industrial and Medical Nuclear Safety
Office of Nuclear Material Safety and Safeguards |
| Technical Contact: |
Robert L. Ayres, NMSS
(301) 415-5746
E-mail: rxa1@nrc.gov |
ATTACHMENT 1
Guidance to Users of Nycomed Amersham and North American Scientific
(NAS), Inc. I-125 Interstitial Sources
Dosimetry and Calibration Changes
This guidance is to alert physicists and radiation oncologists to two changes affecting calculations of dose from 125I interstitial brachytherapy sources: (a) The National Institute of Standards and Technology (NIST) is implementing a new primary calibration standard for these sources on 1 January 1999. Sources marketed in 1998 will undergo a change in calibration on 1 January 1999. New source designs that become available after this date will be calibrated to the new standard; (b) In 1995, the American Association of Physicists in Medicine (AAPM) published a revised dose-calculation protocol, developed by its Task Group 43 and known as the TG43 report. Both of these changes require modification of the dosimetric constants stored in the input files of radiation treatment planning (RTP) software and may involve revising the prescribed dose. Following the protocol herein will not modify doses delivered to patients . The TG43 protocol and the revised NIST standard, implemented either singly or in combination, simply provide a more rigorous estimate of the absorbed dose that brings calculated stated doses into closer alignment with doses actually delivered to patients. Physicists should not implement these changes without consulting with the responsible radiation oncologist. Failure to modify dose-calculation procedures in response to the new calibration standard will result in dose-delivery errors, relative to past practice, of 10% while incorrect application of these factors can result in 20% or even 30% errors. This notice briefly reviews the procedure for adapting to these changes: more detail are given elsewhere2.
Revised Low-Energy Seed Calibration Standard
On January 1, 1999 NIST will implement its revised air-kerma strength (SK) standard for low-energy interstitial brachytherapy seeds7. Compared to seeds calibrated prior to this date, calibration values will numerically decrease by about 10%. The affected 125I interstitial sources, which were marketed before January 1, 1999, are the model MED3631-A/M source, currently marketed by North American Scientific (NAS) Corporation, the currently unavailable model MED3631-A/S (the predecessor of model MED3631-A/M) source and the Nycomed Amersham models 6711 and 6702 sources. Up to January 1, 1999, these sources were calibrated against the prior NIST SK standard implemented in 19854, Other products containing Model 6711 seeds (Models 6720 and 7000) are affected as well. To avoid confusion, air-kerma strength traceable to the 1985 and 1999 standards will be denoted by SK,85std and SK,99std, respectively. Both quantities have units of µ×m2×h-1. Although NIST will implement the new standard on 1 January 1999, immediate implementation of SK,99std calibrations by vendors can not be guaranteed. For seeds shipped after 1 January 1999, users should check with the vendor to identify the standard to which their calibrations are referenced.
SK,85std calibrations were based upon Loftus' air-kerma measurements, made using the Ritz low-energy free air chamber (FAC) which officially became the primary standard in 19854. These measurements were affected by 4.5 keV titanium characteristic x-rays, the significance of which was not appreciated at the time. The net effect of these low-energy x-rays, which do not contribute to dose in water at distances beyond 1 mm, was to inflate all SK,85std values traceable to these measurements by about 10% relative to the penetrating component of the photon spectrum9. SK,99std calibrations are based upon measurements using Loevinger's wide-angle free-air chamber (WAFAC) with a thin absorber to eliminate the Ti x rays7. For all three model seeds (6711, 6702, and 3631A/S) SK,85std and SK,99std are related by:
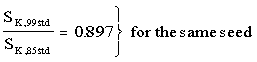
(1)
Task Group 43 Dosimetry Revisions
In 1995, the AAPM issued its Task Group 43 (TG43) report on interstitial brachytherapy dosimetry5. The TG43 report included a new single-source dose-calculation formalism and recommended dosimetry constants for models 6711 and 6702 sources based upon TLD dose-rate measurements normalized to SK,85std. Subsequently, TLD dose-rate measurements normalized to SK,85std were reported for the NAS model 3631A/S source8. Similar measurements for the MED3631-A/M source have been performed but are not yet published. In the TG43 formalism, the connection between absolute dose rate and source strength is given by the dose-rate constant, 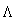 TG43,85std ( = dose rate in water at 1 cm/SK,85std).
TG43,85std ( = dose rate in water at 1 cm/SK,85std). 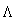 TG43,85std deviates (see table I) by 10%-18% from the dose-rate constant,
TG43,85std deviates (see table I) by 10%-18% from the dose-rate constant, 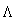 preTG43,85std derived from the most widely used dose-distribution data available prior to the TG43 report3. Most of this discrepancy is due to inflation of SK,85std by low-energy photons: the pre-TG43 dose-calculation models assume that all photons contributing to SK also contribute to tissue-medium dose rates at distances up to several cm. In contrast, the TG-43 data are based upon dose measurements in a phantom which are not affected by the low energy photons
preTG43,85std derived from the most widely used dose-distribution data available prior to the TG43 report3. Most of this discrepancy is due to inflation of SK,85std by low-energy photons: the pre-TG43 dose-calculation models assume that all photons contributing to SK also contribute to tissue-medium dose rates at distances up to several cm. In contrast, the TG-43 data are based upon dose measurements in a phantom which are not affected by the low energy photons
Table I: Dose-rate constants, 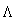 , for I-125 interstitial sources
, for I-125 interstitial sources
(Units of cGy×h-1×U-1 or cGy×h-1/(µ×m2×h-1)
| Source Model |
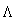 preTG43,85std preTG43,85std |
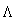 TG43,85std TG43,85std |
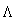 TG43,99std TG43,99std |
| 6711 |
1.039 |
0.88 |
0.98 |
| 6702 |
1.039 |
0.93 |
1.04 |
| MED3631-A/S |
---- |
0.93 |
1.04 |
| MED3631-A/M |
---- |
----† |
----† |
†Publication of measurements pending: contact NAS for most current estimates of TG43 dosimetry ratios.
Recommended AAPM Implementation Plan
To simplify clinical implementation of these changes, the AAPM recommended a two-stage clinical implementation plan2: (a) First implement the TG43 dosimetry protocol for I-125 seeds; and (b) then adopt the revised air-kerma strength standard. Implementation of TG43 dosimetry means modifying the dosimetry constants stored in the appropriate RTP input file so that the predicted dose rates per unit strength agree with the dose-rate distributions recommended by the report. Such implementation is straightforward for Radiation Treatment Planning (RTP) systems that use the TG43 dosimetric ratios to input dose distributions. Implementation is more complicated, but still achievable, for systems using older dose-calculation formalisms2.
(a)Task Group 43 Implementation
The TG43 formalism predicts dose rates that are smaller by 10-18% than their pre-TG43 counterparts due mostly to changes in the dose-rate constant, 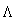 , and anisotropy constant,
, and anisotropy constant,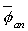 , Thus, using TG43 dose calculations for clinical treatment without corresponding changes in the prescribed dose, DPx , will abruptly increase doses actually delivered to patients by 10-18%. To avoid this scenario, physicists should compare the doses calculated by their pre-TG43 algorithm, DpreTG43,85std, to those calculated from the TG43 report for an identical geometry. Then, the prescribed dose, DPx,TG43, used to determine the required source strength in conjunction with the TG43 dose distribution should be adjusted as follows:
, Thus, using TG43 dose calculations for clinical treatment without corresponding changes in the prescribed dose, DPx , will abruptly increase doses actually delivered to patients by 10-18%. To avoid this scenario, physicists should compare the doses calculated by their pre-TG43 algorithm, DpreTG43,85std, to those calculated from the TG43 report for an identical geometry. Then, the prescribed dose, DPx,TG43, used to determine the required source strength in conjunction with the TG43 dose distribution should be adjusted as follows:
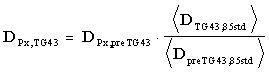
(2)
where the brackets represent spatial averaging over the typical implant geometry. Bice1 et al. have shown that for prostate implants consisting of Model 6711 seeds planned with the isotropic point-source model using the most commonly used pre-TG43 dosimetry constants (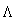 preTG43,85std = 1.039 and = 0.87)3 and the corresponding TG43 values (
preTG43,85std = 1.039 and = 0.87)3 and the corresponding TG43 values (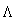 TG43,85std = 0.88 and = 0.93), the prescribed dose correction factor is
TG43,85std = 0.88 and = 0.93), the prescribed dose correction factor is
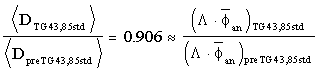
(3)
independently of the detailed averaging procedure. In this example, the typical prescribed dose of 160 Gy for definitive treatment of prostate cancer by permanent I-125 seed implant alone should be lowered to 144 Gy. Although equation (3) represents a common scenario, it is essential that each institution base their dose prescription adjustment on a comparison of their own pre-TG43 dose calculations to the appropriate TG43 implementation. Readers are warned that the appropriate dose prescription factor for their clinical application depends on the dose calculation model and dosimetry constants historically used in their practices, the seed model, and the implant geometry. The example described above is not valid for all implant types and dose-calculation models. Failure to adjust the prescribed dose upon adopting the TG43 dosimetry calculations will result in an unintended increase in dose delivered to the patient as large as 18%. Adjusting the prescribed dose in accord with equation (2) will not change doses actually delivered to patients, but will eliminate differences between calculated dose and dose actually delivered to the patient relative to TG43 calculations.
To reduce the possibility of errors or misunderstandings, physicists should not implement revised dose prescription guidelines nor implement TG43 dose calculations without consulting with the responsible radiation oncologist. The revised dose calculations should be verified and all involved staff trained prior to treating patients with the new protocol. Implementation of dose calculations numerically equivalent to TG43 recommendations on RTP systems not supporting the TG43 formalism is discussed in reference 2.
(b)Adapting to the 1999 NIST standard, SK,99std following TG43 implementation
For all patients treated with I-125 seeds specified in terms of air-kerma strength traceable to the 1999 NIST standard, the dose-rate constant used by the treatment planning system should be updated to the value 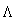 TG43,99std given in table I. This modification will not change the dose delivered to the patient because the source strength values entered into the planning system will be correspondingly lower than those used with the 1985 standard. No revision of the prescribed or stated dose is necessary. Failure to revise the dose-rate constant appropriately will result in erroneous dose delivery. Following the dose-rate constant update but before treating patients, a physicist should verify that this change uniformly increases dose rates predicted by the treatment planning system by 11.5% for implants with numerically identical SK values before and after the revision. This recommendation applies only to the Models 6711, 6702, MED3631-A/S and MED3631-A/M seeds; recommended dosimetry constants for new interstitial products entering the market after 1 January 1999 will already be normalized to the new source-strength standard. However, for the MED3631-A/M seed, additional dose-rate measurements will soon be available for review. Users of this product may want to consider adopting updated TG43 dosimetric ratios at the same time they adopt the revised NIST air-kerma strength standard. The vendor, North American Scientific, should be contacted for further information.
TG43,99std given in table I. This modification will not change the dose delivered to the patient because the source strength values entered into the planning system will be correspondingly lower than those used with the 1985 standard. No revision of the prescribed or stated dose is necessary. Failure to revise the dose-rate constant appropriately will result in erroneous dose delivery. Following the dose-rate constant update but before treating patients, a physicist should verify that this change uniformly increases dose rates predicted by the treatment planning system by 11.5% for implants with numerically identical SK values before and after the revision. This recommendation applies only to the Models 6711, 6702, MED3631-A/S and MED3631-A/M seeds; recommended dosimetry constants for new interstitial products entering the market after 1 January 1999 will already be normalized to the new source-strength standard. However, for the MED3631-A/M seed, additional dose-rate measurements will soon be available for review. Users of this product may want to consider adopting updated TG43 dosimetric ratios at the same time they adopt the revised NIST air-kerma strength standard. The vendor, North American Scientific, should be contacted for further information.
Adapting to the 1999 NIST standard, SK,99std without adopting the TG43 Report Recommendations.
If the TG43 dosimetry recommendations (or their numerical equivalents in older dose-calculation formalisms) have not been adapted, it is still necessary to modify the constants used by the treatment planning system because of the new 1999 NIST standard. Failure to make the recommended changes will result in an unintended 11.5% increase in doses delivered to patients. The pre-TG43 dose-rate constant should be modified as follows:
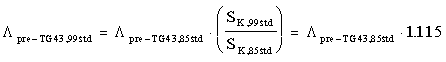
(4)
Implementation of (4) will not require adjustment of the prescribed or stated dose and will not modify doses actually delivered to patients. However, the discrepancy between the institution's calculated doses and doses as determined by the TG43 recommendations will remain unchanged.
Modification of Source-Strength Verification Procedures
With the revision of the air-kerma strength standard, corresponding adjustments must be made to the calibration factors used with dose calibrators or other re-entrant ionization chambers to verify vendor calibrations. To verify seed calibrations traceable to the new standard, multiplicative calibration factors, NSk,85std, used to convert instrument readings into source strengths, SK,85std, should be modified as follows:
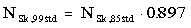
(5)
The product of the instrument reading, the revised factor NSk,99std, and other corrections independent of the calibration standard (e.g., temperature-pressure corrections), will now represent source strength, SK,99std, traceable to the revised standard. This recommendation applies to all seeds marketed before January 1, 1999, including model MED3631-A/M. Calibration factors traceable to the new I-125 standard will be available from the Accredited Dosimetry and Calibration Laboratories. Further guidance in implementing procedures for source-strength verification is given in reference 6.
Whom to Contact For Further Assistance
If you have questions on implementing the TG43 dosimetry recommendations or adapting to the revised source strength standard, please contact the Radiological Physics Center (RPC) at MD Anderson Cancer Center, Houston, TX at (713)-792-3233.
AAPM Ad Hoc Subcommittee on Low-Energy Seed Dosimetry
Jeffrey F. Williamson, Ph.D., Washington University, St. Louis, MO, Chair
Bert M. Coursey, Ph.D., NIST, Gaithersburg, MD
Larry A. DeWerd, Ph.D., University of Wisconsin, Madison, WI
William F. Hanson, Ph.D., M. D. Anderson Cancer Center, Houston, T X
Ravinder Nath, Ph.D., Yale, New Haven, CT
Geoffrey Ibbott, Ph.D., University of Kentucky, Lexington, KY
References
- Bice, W.S., Prestlidge, B.R., Prete, J.J., and Dubois, D.F., "Clinical impact of implementing AAPM Task Group 43 on permanent prostate brachytherapy using I-125," Int. J. Radiation Oncology. Biol. Phys. 40: 1237-1241, 1998
- Kubo, H.D., Coursey, B. M., Hanson, W.F., Kline, R. W., Seltzer, S. M., Shuping, R.E. and Williamson, J. F., "Report of the Ad Hoc Committee of the AAPM Radiation Therapy Committee on 125I Sealed Source Dosimetry", Int. J. Radiation Oncology. Biol. Phys. 40: 697-702 1998
- C.C. Ling, D. York, I.J. Spiro, D. Kubiatowicz, and D. Bennett, "Physical dosimetry of 125I seeds of a new design for interstitial implant," Int. J. Radiation Oncology Biol. Phys. 9, 1747-1752 (1983).
- Loftus, T.P., "Exposure standardization of 125I seeds used for brachytherapy," J. Res. Nat. Bur. Stand. 89, 295-303 (1984).
- Nath, R., Anderson, L.L., Luxton, G., Weaver, K.A., Williamson, J.F. and Meigooni, A.S., "Dosimetry of interstitial brachytherapy sources: Recommendations of the AAPM Radiation Therapy Committee Task Group No. 43", Med. Phys. 22: 209-234, 1995.
- Nath, R., Anderson, L.L., Meli, J.A., Olch, A.J., Stitt, J.A. and Williamson, J.F., "Code of Practice for Brachytherapy Physics: Report of the AAPM Radiation Therapy Committee Task Group No. 56", Med. Phys. 24: 1557-1648, 1997.
- Seltzer, S.M., Lamperti, P.J., Loevinger, R., Soares, C.G., and Weaver, J.T., "New NIST air-kerma strength standards for I-125 and Pd-103 brachytherapy seeds," Poster presentation, AAPM Annual Meeting, August 1998, San Antonio, TX (Abstract: Med. Phys. 25: A170, 1998).
- Wallace, R.E. and Fan, J.J., "Evaluation of new brachytherapy iodine-125 source by AAPM TG43 formalism," Med. Phys. 25: 2190-2196, 1998.
- Williamson, J.F., "Monte Carlo evaluation of specific dose constants in water for 125I seeds," Med. Phys. 15, 686-694 (1988).
ATTACHMENT 2
| Date: |
Tue, Jan 5, 1999 |
| Subject: |
NIST 1999 Calibrations for Iodine-125 |
Dear Colleagues:
This is a brief status report from NIST on air kerma strength measurements for iodine-125 brachytherapy seeds. It is now past January 1, 1999 and NIST will only provide I-125 seed calibrations in terms of air-kerma strength based on the Loevinger WAFAC. We are in contact with all of the announced manufacturers of I-125 seeds and they will, in due course, receive certificates for seeds based on the new WAFAC calibration. Here are some answers to questions that we have been receiving from several quarters:
1) When will a given manufacturer make the transition to the 1999 standard?
That question must be addressed to the manufacturer. It will depend on the schedule for shipping seeds to NIST, the time required for appropriate measurements at NIST, and the time required by the manufacturer to document the change and reflect this on their certificates and shipping papers.
2) What are the implications for the hospital physicists?
NIST will not address this question; all enquiries are directed to the Radiological Physics Center, as recommended by the Guidance Document from the AAPM ad hoc Task Group.
3) Will the ADCLs be able to offer instrument calibrations for the new I-125 seeds?
Wisconsin and K&S are accredited to offer 1999 calibrations for Nycomed-Amersham Model 6702 and 6711 Seeds, and NAS IoGold MED3631 A/S. M.D. Anderson has completed a proficiency test on these same seeds, but has not been accredited by AAPM. NIST does not consider it necessary for the ADCLs to demonstrate proficiency on each new I-125 seed. However, they will need to receive seeds directly from NIST in order to transfer the NIST WAFAC calibration to the ADCL transfer instrument. NIST is asking the manufacturers to supply five seeds for each new seed design. They are also being asked if they would like for us to forward a set of these seeds to the ADCLs to effect a transfer of the national standard to the AAPM network. (The manufacturer is under no obligation to do this.) After the measurements some of these seeds (probably three) will be sent to the ADCLs in the order specified by Dr. Ibbott.
I hope this answers some of the immediate questions, and that we can work together to make this transition to the new NIST air-kerma standard as smooth as possible. I would like to thank everyone for the efforts that they are putting into this, put especially Jeff Williamson for guiding the
preparation of the AAPM Guidance Document and Paul Lamperti for extraordinary efforts to measure seeds over the past six months, including the recent holidays.
Best regards,
Bert Coursey
(NUDOCS Accession Number 9901190183)
Page Last Reviewed/Updated Tuesday, March 09, 2021